Volunteer/Request Ambassador
.svg)
Clinical Trials

Clinical Trial Highlights
Studies are listed in order of study start date.
A Study of Gefitinib, Trametinib, Disulfiram, and Sunitinib in Addition to Standard Chemotherapy in People With Osteosarcoma (OsteEvo)
Sponsor: Memorial Sloan Kettering Cancer Center
Therapy: Methotrexate, Doxorubicin, Cisplatin, Gefitinib, Trametinib, Disulfiram, Sunitinib, Ifosfamide, and Biopsy.
Study Start: March 2026
Phase 2
Contact: reedd1@mskcc.orgAtezolizumab in Combination With Stereotactic Body Radiation Therapy (SBRT) and Surgery for Relapsed Osteosarcoma
Sponsor: Emory University
Therapy: Atezolizumab, SBRT
Study Start: March 2026
Phase 1
This early-phase clinical trial is testing a combination of immunotherapy (atezolizumab), targeted radiation, and surgery for patients whose osteosarcoma has returned in the lungs. The goal is to find a safe way to use these treatments together and see if they may better control the cancer compared to current approaches. Patients up to 50 years old are eligible.
Contact: aflacdevtreferral@choa.orgA Phase 2 Trial of Zanzalintinib in Advanced/Metastatic Bone Sarcomas (ZAMBONE)
Sponsor: M.D. Anderson Cancer Center
Therapy: Zanzalintinib
Study Start: February 2026
Phase 2
This Phase 2 study is testing an oral drug called zanzalintinib in adults with advanced bone cancers, including osteosarcoma, that have spread or returned. Researchers are checking if this new treatment can stop tumor growth in patients who have already tried standard therapies.
Contact: jalivingston@mdanderson.orgLu-TARGO (177Lu-TARGeted Osteosarcoma Therapy)
Sponsor: Lantheus Medical Imaging
Therapy: LNTH2403 Phase 1 dose
Study Start: February 2026
Phase 1/2
This trial is testing a new lutetium-labelled monoclonal antibody radiotherapeutic called LNTH-2403 for patients with osteosarcoma that has come back or stopped responding to standard chemotherapy. Eligible participants are generally aged 12 and older (18+ for the initial phase), weigh at least 30 kg, and must have already gone through at least one round of chemotherapy.
Contact: clinicaltrials@lantheus.comGenetically Engineered Cells (FH-FOLR1 ST CAR T Cells) for the Treatment of Advanced Refractory or Recurrent/Progressive Osteosarcoma, FIERCe Trial
Sponsor: Fred Hutchinson Cancer Center
Therapy: FH FOLR1 ST CAR T-cells
Study Start: January 2026
Phase 1
This early-phase clinical trial is testing a new type of immunotherapy called CAR T-cell therapy targeting the FOLR1 protein on tumour cells, where a patient’s own immune cells are collected, reprogrammed in a lab to recognise osteosarcoma, and then given back to help attack the cancer. Ages 1-75 are eligible and must have evaluable or measurable disease.
Contact: immunotherapy@fredhutch.orgTegavivint With Gemcitabine in Patients With Relapsed or Refractory Osteosarcoma
Sponsor: Emory University
Therapy: Tegavivint, Gemcitabine
Study Start: January 2026
Phase 1
This early-phase clinical trial is testing a new drug (tegavivint) in combination with chemotherapy (gemcitabine) for patients whose osteosarcoma has come back or not responded to treatment. The main goal is to find a safe dose and understand side effects, while also seeing if this combination may help slow or shrink the cancer. Ages 1-30 are eligible.
Contact: aflacdevtreferral@choa.orgDFMO Maintenance for Patients With Relapsed/Refractory Ewing Sarcoma or Osteosarcoma
Sponsor: Montefiore Medical Center
Therapy: DFMO
Study Start: April 2025
Phase 1/2
This clinical trial is studying a drug called DFMO as a “maintenance therapy” for patients with osteosarcoma whose cancer has come back but is not currently detectable after treatment, with the goal of helping prevent it from returning. Researchers are mainly looking at whether this approach is safe and doable and whether it may help keep the cancer from coming back longer. Patients up to 39 years old are eligible.
Contact: rzylber@montefiore.orgIscador® P (Mistletoe) Immunotherapy for Recurrent Osteogenic Sarcoma
Sponsor: Hackensack Meridian Health
Therapy: Iscador® P
Study Start: May 2024
Phase 2
This clinical trial is testing an immunotherapy made from mistletoe extract (Iscador® P) for patients with osteosarcoma that has come back in the lungs but has been completely removed with surgery. The goal is to see if this treatment can help keep the cancer from returning longer after surgery by supporting the body’s immune system. Patients ages 8-30 are eligible.
Contact: laura.goode@hmhn.orgAn Organoid-based Functional Precision Medicine Trial in Osteosarcoma (PREMOST)
Sponsor: Jonsson Comprehensive Cancer Center
Therapy: Functional Drug Testing
Study Start: February 2024
This clinical trial is studying whether doctors can use a patient’s tumor sample to grow “mini tumors” in the lab and test different chemotherapy drugs on them to see which ones might work best. The goal is to learn if this approach could help personalize treatment in the future by identifying which therapies are most likely to be effective for each individual patient. All ages are eligible.
Contact: summer.norris@cuanschutz.eduAllogeneic Expanded Gamma Delta T Cells With GD2 Chemoimmunotherapy in Relapsed /Refractory Neuroblastoma or Refractory/ Relapsed Osteosarcoma
Sponsor: Emory University
Therapy: Ex Vivo Expanded Allogeneic γδ T Cells in Combination with Dinutuximab, Temozolomide, Irinotecan and Zoledronate Study
Start: November 2023
Phase 1
This early-phase clinical trial is testing a combination of donor immune cells (called gamma delta T cells) with chemotherapy and an antibody drug to treat osteosarcoma that has come back or is not responding to treatment. It is open to patients 12 months and older and is focused on finding a safe dose and understanding how well this approach might work.
Study to Assess Safety and Efficacy of Vactosertib in Adolescents and Adults With Recurrent, Refractory or Progressive Osteosarcoma
Sponsor: MedPacto, Inc.
Therapy: Vactosertib
Study Start: May 2023
Phase 1/2
This early-phase clinical trial is testing a new oral drug called vactosertib for people with osteosarcoma that has come back or is not responding to treatment, to see if it is safe and whether it may help slow or shrink the cancer. It is open to adolescents and adults ages 12 and older with relapsed or refractory disease.
Contact: Kristen.VanHeyst@UHhospitals.orgAtezolizumab and Cabozantinib for the Treatment of Adolescents and Young Adults With Recurrent or Metastatic Osteosarcoma, TACOS Study
Sponsor: MD Anderson Cancer Center
Therapy: Atezolizumab, Cabozantinib
Study Start: April 2023
Phase 2
This phase 2 clinical trial is testing a combination of two drug —atezolizumab (an immunotherapy) and cabozantinib (a targeted therapy)—for people with osteosarcoma that has come back or spread after standard treatment, to see if it can help control the cancer. It is open to adolescents and young adults ages 12 and older whose disease is recurrent or resistant to prior therapies.
Contact: jalivingston@mdanderson.orgA Study to Test the Addition of the Drug Cabozantinib to Chemotherapy in Patients With Newly Diagnosed Osteosarcoma
Sponsor: National Cancer Institute (NCI)
Therapy: MAP + cabozantinib
Study Start: March 2023
Phase 2/3
This phase 2/3 clinical trial is testing whether adding a targeted drug called cabozantinib to standard chemotherapy at diagnosis may work better than chemotherapy alone for people with newly diagnosed osteosarcoma. It is open to children, adolescents, and young adults up to age 40 who are newly diagnosed and have not yet started treatment.
Contact: oncologyresearch@peds.uab.eduEx Vivo Drug Sensitivity Testing and Multi-Omics Profiling
Sponsor: Florida International University
Therapy: Functional Drug Testing
Study Start: February 2023
This clinical trial is studying a personalized approach where a patient’s tumor sample is tested in the lab against different cancer drugs to help identify which treatments might work best for that individual. It is open to children and young adults up to age 21 with cancers that have come back or are not responding to treatment, and requires available tumor tissue from a recent surgery or biopsy. Patients may enroll remotely.
Contact: fpmlab@fiu.eduA Study of Cabozantinib as a Maintenance Agent to Prevent Progression or Recurrence in High-Risk Pediatric Solid Tumors
Sponsor: Nationwide Children's Hospital
Therapy: Cabozantinib
Study Start: July 2022
Phase 2
This clinical trial is studying a drug called cabozantinib as a maintenance therapy to help prevent cancer from coming back or growing after initial treatment in patients with high-risk osteosarcoma and other similar cancers. It is open to children, adolescents, and young adults ages 18 months to 40 years who have already responded to prior treatment and have little or no remaining detectable disease.
Thoracotomy Versus Thoracoscopic Management of Pulmonary Metastases in Patients With Osteosarcoma
Sponsor: Children's Oncology Group
Therapy: Surgery
Study Start: April 2022
Phase 3
This phase 3 clinical trial is comparing two different types of lung surgery—traditional open surgery versus a minimally invasive approach using small incisions—to see which works better for patients with osteosarcoma that has spread to the lungs. It is open to children, adolescents, and adults up to age 50 who have a limited number of lung tumours that can be safely removed with surgery.
Contact: Find a contact at one of the 232 study locationsLosartan + Sunitinib in Treatment of Osteosarcoma
Sponsor: University of Colorado, Denver
Therapy: Losartan, Sunitinib
Study Start: August 2019
Phase 1
This early-phase clinical trial is testing a combination of two oral drugs (losartan and sunitinib) to see if they are safe and whether they may help slow the growth of osteosarcoma that has come back or not responded to treatment. It is open to patients ages 10 and older who have relapsed or refractory disease after at least one prior therapy.
Contact: kelly.faulk@childrenscolorado.orgStudy of CAR T-Cells Targeting the GD2 With IL-15+iCaspase9 for Relapsed/Refractory Neuroblastoma or Relapsed/Refractory Osteosarcoma
Sponsor: UNC Lineberger Comprehensive Cancer Center
Therapy: iC9.GD2.CAR.IL-15 T-cells
Study Start: February 2019
Phase 1
This early-phase clinical trial is testing a type of immunotherapy called CAR T-cell therapy, where a patient’s own immune cells are modified in a lab to better recognize and attack osteosarcoma that has come back or not responded to treatment. It is open to patients ages 1 and older with relapsed or refractory disease, and is focused on finding a safe dose and understanding how well this approach may work.
Contact: UNCImmunotherapy@med.unc.eduLiquid Biopsy in Ewing Sarcoma and Osteosarcoma as a Prognostic And Response Diagnostic: LEOPARD
Sponsor: Dana-Farber Cancer Institute
Therapy: FoundationOne Liquid CDx (liquid biopsy)
Study Start: May 2018
Phase 1
This clinical trial is studying a liquid biopsy blood test that looks for tiny pieces of tumour DNA in the bloodstream to help predict whether osteosarcoma might come back and how patients are responding to treatment. It is open to children, teens, and adults ages 12 months to 50 years who are newly diagnosed and just starting standard chemotherapy and mainly involves giving blood samples over time rather than testing a new treatment.
Contact: david_shulman@dfci.harvard.eduLearn About Clinical Trials
Clinical trials play a critical role in improving osteosarcoma care by helping researchers better understand which treatments are most effective and for whom. The therapies available today exist because patients and families chose to take part in research, contributing to that progress in meaningful ways. For those facing osteosarcoma, a clinical trial may offer access to new treatment approaches along with close medical oversight, while also helping move the field forward for others. At MIB Agents, the goal is to provide clear, reliable information to support thoughtful, informed conversations between patients and their care teams about available options.
Why Should I Consider a Clinical Trial? (NCCN)Questions to Ask Before joining a clinical trial (FDA)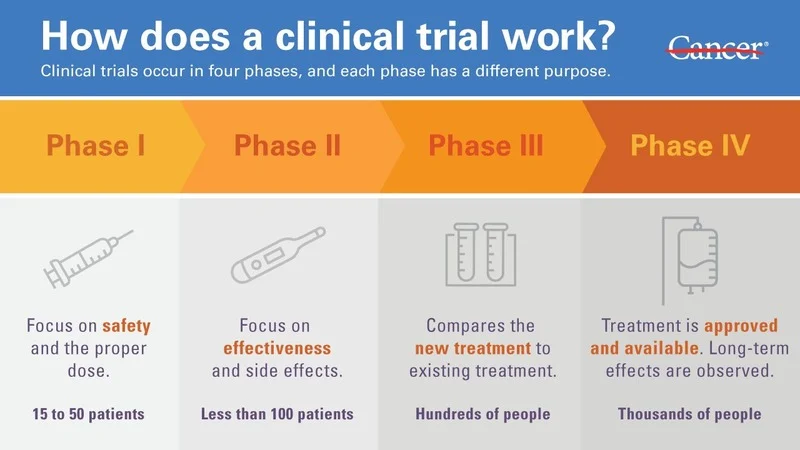
Explore Clinical Trial Webinars on OsteoBites
Patient Support Resources for Clinical Trials

Clinical Trials Assistance
This programme assists with direct expenses related to transportation (airfare, gas, rental cars, taxi fare, parking/tolls), and lodging associated with participation in a clinical trial.
Clinical Trial Navigator
Need help with your search or connecting with trial sites? Request a contact to speak with a Clinical Tria Navigator today.
Clinical Trial Glossary
Clinical Trial Status
Before enrolling its first patient, the study was stopped.
These clinical trials have finished normally and treatment and/or follow-up visits have ended.
These clinical trials have ended early and will not start again. When a clinical trial is terminated, it means participants stop being treated or followed.
These clinical trials have been stopped early but they may start again.
These clinical trials are already underway and new participants are not being enrolled.
These studies are not open to the general public even if a person meets all eligibility criteria. Instead, a select group of patients determined by the researchers are invited to join the trial.
Patients who meet eligibility criteria are currently being enrolled in these clinical trials
The researchers have not yet started recruiting patients for these clinical trials
Study Design
Groups of participants are treated with multiple interventions in a specific order. For instance, participants in one group will receive drug A first and drug B second, while participants in another group with receive drug B first and drug A second.
Participants do not receive any intervention or treatment.
Participants receive a device or procedure that is made to look the same as the device or procedure that is being studied in the clinical trial, but does not have any active components
Participants receive a placebo, a treatment that has no therapeutic effect.
Participants receive a type of treatment that is already known to be effective
Participants receive the treatment that is the subject of the clinical trial.
A group or subgroup of participants in a clinical trial (usually an interventional study) that receives the same intervention or no intervention, consistent with the study protocol.
A group of participants who share the same characteristics. Cohorts are often used in observational studies.
A type of clinical study design where researchers observe participants but do not control the treatment they receive. These types of studies are usually done when it is difficult or unethical to randomly assign participants to experimental groups.
A type of clinical study design that allows researchers to test a potential treatment (or intervention) in participants.
The process used to make participants in clinical trials aware of potential risks and benefits of participation.
An adolescent’s agreement to participate in a clinical trial. Before giving assent, the clinical trial must be explained in a way the child can understand. Additionally, both parents need to permit participation.
Inclusion Criteria
Cancer treatment that targets the entire body. Chemotherapy is the most common form of Systemic cancer therapy.
Cancer that has not grown or spread past the primary site.
Cancer that has spread from where it started (call the primary site) to other parts of the body.
Cancer is not responding to treatment.
Cancer has returned after treatment and after a period where no cancer was detected.
Cancer or tumor that can be removed completely with surgery
Cancer or tumor that cannot be removed completely with surgery
Outcomes
Used to measure deaths from all causes that occurred during a clinical trial.
Any unwanted medical occurrence that is considered life-threatening, leads to hospitalization or increases existing hospitalization, disrupts the ability to conduct normal life functions, leads to congenital anomalies or birth defects, or results in death.
Any unwanted medical occurrence that takes place during a clinical trial or within the follow-up period. These events may or may not be caused by participation in the clinical trial.




